Difference between revisions of "Intercrystolecular snapping modes"
m (→Bend snapping) |
(→Bend snapping – and other snapping modes: added illustrative image) |
||
| Line 71: | Line 71: | ||
== Bend snapping – and other snapping modes == | == Bend snapping – and other snapping modes == | ||
| + | |||
| + | [[File:Crystolecule-vdW-questions snapping-bend-n-more.png|400px|thumb|right|'''Bend-snapping:''' Energy of bulk deformation gets unconstrained released. This can be a huge amount. '''Stick-slip-snapping:''' A combination of high pressure notch-snapping and bend-snapping. '''Surface bond snapback:''' Just surface atom bons(s) snap back. '''Entropic snapping:''' The snap is caused by an entropic force as a motion in a certain direction increases the thermal degrees of freedom (more micro-states define a more likely future). This may be significant.]] | ||
'''A [[crystolecule]] part is bent and then suddenly released without any back-holding force.''' <br> | '''A [[crystolecule]] part is bent and then suddenly released without any back-holding force.''' <br> | ||
| Line 87: | Line 89: | ||
* can easily be >> vdW energy >>> notch energy | * can easily be >> vdW energy >>> notch energy | ||
(all meant per unit area) | (all meant per unit area) | ||
| + | |||
| + | === Stick-slip-snapping === | ||
| + | |||
| + | A combination of high pressure notch-snapping and bend-snapping. | ||
| + | |||
| + | === Surface atom bond snapback === | ||
| + | |||
| + | See page: [[Snapback]] | ||
== Snapback == | == Snapback == | ||
Revision as of 17:58, 25 February 2025
What is considered a to be a "snap" here?
A more or less forced slow motion (nm/s to mm/s) causing
– a sudden removal of motion constraints and following
– a more or less violent fast mechanical relaxation near the speed of sound (many m/s to km/s)
The forced motion can but does not need to provide the majority for the energy for the snap
it can also just release energy that has been stored in the system at an earlier time.
The forced motion must merely provide enough energy to overcome the activation energy for the snap.
(wiki-TODO: add illustrative images)
Contents
[hide]Slide (freely to max contact area) snapping
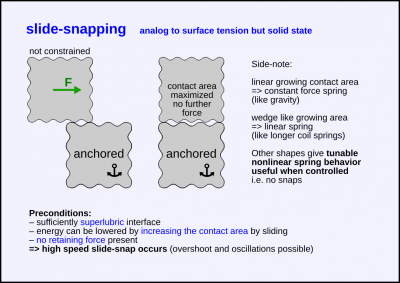
A crystolecule is released while suberlubrically contacting an other crystolecule.
The crystolecule will snap to the location and orientation of maximal surface contact area (surface tension analogy).
Perhaps the most obscure / unexpected one of the snapping modes.
This snapping mode is related to the wedge slide force that should be very useful
for the creation of compact and tunable (constant/linear/nonlinear) weak force springs.
Related: VdW suck-in
Level of energy
- quite weak, less than bend snapping
- likely less than notch snapping in most if not all cases (for same surface area)
=> thus the need for superlubric interfaces for this snapping mode to be available
Notch snapping
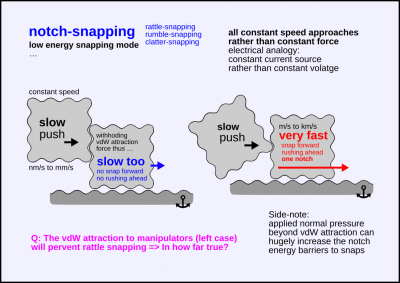
A crystolecule part contacts an other one and snaps to the next notch in commensurate atomic alignment.
I.e. the atomic patterns interdigitate and there's a jump to the next notch due to a shearing force.
Notch snapping can likely be avoidable in assembly and disassembly of structures,
It seems likely that in most cases such snaps are prevented by larger back-holding forces.
In particular face-to-face sticking vdW forces towards the actuating part may prevent notch snapping.
It depends on the ratio of surface areas.
It may even be slightly challenging to intentionally provoke a notch snap.
Cases of weaker back-holding forces that notch snapping forces may be able to overcome:
– shifting a part with merely an edge or a corner of an other part (low attractive vdW forces)
– competition with higher energy notch snapping on a second contacting surfaces that is forcibly moved
– Superbubic sliding to maximize contact area (surface tension analogy)
Level of energy
- low energy snapping mode
Clap snapping
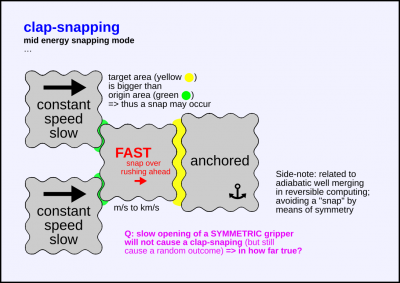
A crystolecule part snaps over from a smaller contact area to a bigger contact area.
Flat on flat like a clap thus the choice of name.
In symmetric cases like with parallel acting gripper claws,
there won't be a snap so long the holding forces before the gripping
exceed uncompensated forces form imperfections in the symmetry of the approach of the gripper.
Side notes:
– Grippers might for the most part not be necessary due to exploitability of vdW sticking forces.
– During symmetric gripper opening without a definite target (in free space or on superlubric surface),
there will be no snap but a one bit of entropy truly random outcome on which side of the gripper the part will end up.
See related page: Well merging and splitting
Level of energy
- mid energy snapping mode
- can possibly be > covalent bond energy given enough area acting
Bend snapping – and other snapping modes
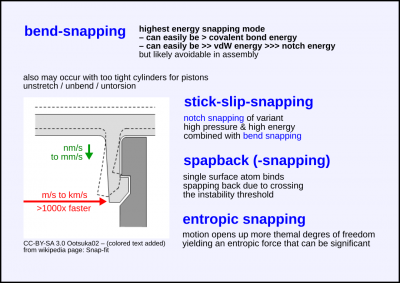
A crystolecule part is bent and then suddenly released without any back-holding force.
Many covalent bonds are stretched and they can be stretched/strained to a high degree.
More than 10% (see page: Superelasticity).
Thus such snaps can release a huge amount of energy.
Bend snapping can likely be avoidable in assembly and disassembly of structures,
it may sometimes be desired, but in most cases weaker energy barriers should easily suffice
i.e. easily prevent thermally activated fall-apart even for the highest expectable temperatures.
Level of energy
- high energy snapping mode
- can easily be > covalent bond energy
- can easily be >> vdW energy >>> notch energy
(all meant per unit area)
Stick-slip-snapping
A combination of high pressure notch-snapping and bend-snapping.
Surface atom bond snapback
See page: Snapback
Snapback
This is sort of surface localized bend snapping.
It can be just single individual covalent bonds that snap back from a bent position
See main page: Snapback
Level of energy
- depends on the number of bonds bent
- much less than bend snapping as it is located on surface only
- much more than notch snapping as the benting of covalent forces is involved, not just redirected vdW forces
Why to avoid snaps (locally and nearby remotely)
- Misplacements during assembly of nano- and microscale parts
- Disturbance of mechanosynthesis (the same energy in non-thermal spectrum can have much higher amplitudes)
- Energy dissipation: Coolability and energy efficiency. Relevant for advanced systems.
The fewer the snaps the higher the assembly speed and throughput can be so long dissipation from snaps is the bottlenecking factor. Especially likely at large nanoscale microscale and lower mesoscale.
Related
- Intercrystolecular interactions
- Intercrystolecular snapping modes < you are here
- Intercrystolecular levitation
- Assembly from atomically precise nanoscale and microscale parts
- Assembly level 2 (gem-gum factory) & Crystolecule assembly robotics & Sequence of zones
- Lagrangian mechanics for nanomechanical circuits – for how violent sharp shocks propagate to the system
- Mechanical-electrical analogies – constant speed sources (in analogy to constant current sources) prevent can prevent undesired snapping
- VdW suck-in – slide snapping
- Accidental heatpump – entropic snap (?)