Nanoscale surface passivation
Old page: Surface passivation


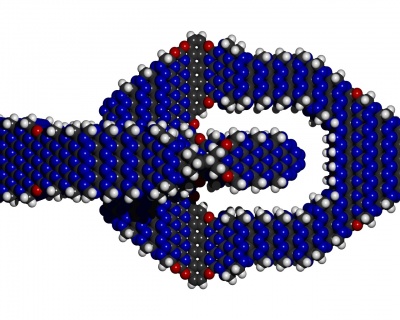
Intuitively passivation is like the flour on the bread-dough that one adds such
that it does not stick together with itself and just about anything.
Also it prevents material surfaces from oxidizing. More on that later.
- Surfaces of crystolecules that are supposed to come in contact with other surfaces need to be passivated in order to not fuse together irreversiby .
- Surfaces of crystolecules that are supposed to come in contact with air need passivation.
(well, first air contact may be used for passivation of non-sliding surfaces but the result may be somewhat random)
Up: Passivation (disambiguation)
(wiki-TODO: eventually merge in the old duplicate page Surface passivation)
Protection against oxidation
This is not as important as it sounds.
Most of the materials that are especially suitable for gemstone metamaterial technology (gemstone like compounds)
are already oxidic in nature, fully odidized, or highly inert towards air.
The whole nanoscale parts (crystolecules) are often made from the same material as a macroscopic (but thin) passivation layer so to say.
Nanoscale passivation is mostly not meant to prevent oxidation but rather meant to:
- prevent irreversible stick together and
- provide smooth sliding surfaces for superlubricity.
Also only a very very small fraction of nano structures in gemstone metamaterial products are exposed to air.
Even less so for moving active nanomachinery.
Most nanostructures are well sealed inside the products.
So the vast majority of nano structures that resides inside the products and nerver comes into contact with air
allows for a much bigger design space in choice of base material and eventual passivation.
Itnuiton: An apple or a babana does not get brown unless you cut them open.
passivation of diamond, lonsdaleite and other allotropes of carbon
Diamond and it's allotropes are best passivated with hydrogen.
This works very well and makes vers stable surfaces. We know that from hydrogarbon chains that make up
gasoline and all of our current day plastics.
Passivation of diamondoid compounds
(wiki-TODO: Add some illustrative images.)
Diamondoid compounds as
- zincblende structure (ABCABC stacking) and
- wurtzite structure (ABAB) structure and
- everyting in-between.
Cubic (111) equivalent to hexagonal (0001) faces
Hydrogen (could be seen as the 0th halogen) works well for passivating cubic diamond (111) faces and equivalents but
due to the small size of hydrogen it has a bit larger gaps and stronger corrugation than hydrogen passivated (110) faces.
Halogens:
True halogens work similarly as hydrogen.
Fluorine (as first halogen) bonds even stronger than hydrogen and smooths tings out a bit more than hydrogen.
Cost being that fluorine makes tings toxic when burnt and in the very long run it is not extremely abundant.
On diamond and lonsdaleite (and likely sp3 boron nitrides) these two elements (H & F) are special in that
they can withstand quite severe chemical attacks and are thus good environment facing surfaces.
Chlorine (as the second halogen) bonds weaker than hydrogen.
This may vary with the context of different chemical attacks.
Burning products still toxic but maybe not quite as bad as fluorine.
Bigger atoms more smooting.
Bromine and iodine (as the last two halogens) are still weaker bonding.
This gets quite fragile, especially to UV light.
Diminising returns in growing atom size and thus in surface smoothing.
The higher period the base element(s) the bigger the atoms and
the bigger the corrugation and the weaker the bonding. Some exceptions not all entirely monotonous.
Biggest step is from carbon to silicon, further to germanium and sp3 grey tin
atom sizes grow slower bit bonding quickyl weakens.
Lead as pure element does not even form diamondoid sp3 structures anymore.
Similar considerations hold for III-V compounds.
Calcogens with their typical two bonds mismatch the threefold symmetry of (111) faces.
OH, SH, SeH groups can be added getting rapidly more fragile (and toxic, not as bad as F and maybe Cl).
Pnictogens (N,P,As,Sb,(Bi)) seem to be an awesome match for cubic(111) or equivalent surface passibation.
Not as additions onto but as replacements of the topmost layer of base material atoms.
This gets completely rid of the singly bonded passivating halogen atoms replacing them with the lone pairs of the pnictogens.
The advantage being quite a bit smoother surfaces and aversion of snapback isntabilities.
As bond stretching is 20x stiffer than bond bending.
(TODO: Find out in how far a lone pair can be bent like a bending a bond to an atom.)
The disadvantage is that strain is added to the surface as
the included atoms do not perfectly match the size of the base material (in average).
So a compensating opposing passivated side (or very deep material) is needed (unless a bending is actually a desired effect).
Going up the periods chemical reactivity becomes increasingly of concern.
Matching the same periods seem self suggesting to mimimize stress and strain(i.e. bending).
Environment exposure is a much more constraining condition than mutual exposure under vacuum for a sliding interface.
Nitrogen N passivation of Si(111) seems quite plausible and even chemically stable.
Aside an quite intense dipol moment of such surface that could maybe even be used for
exotic repulsive Keesom forces (the real dipole to real dipol forces part of the van der Waals forces).
Phosporus P passivation off silicon may be quite reactive to even water liking to become phoshoric acid (H3PO4)
But in a vaccuum mutually contactin it might work just fine judging from tings like phosphorene:
https://en.wikipedia.org/wiki/Phosphorene
Arsenic passivation of germanium it gets mre sketchy (and toxic).
Antimony passivation of grey tin … wild.
Bismuth like lead is no longer forming sp3 structurs
(single atoms could likely be coaxed into submission)
With the bigger atoms not so different in size anymore
there is more freedom in cross combination without introducing as much stresses and strains.
Binary compounds could have atching binary passivations.
Generally a lot over averaging tricks can be played over slightly larger scales.
Cubic (100) faces
For diamond hydrogen or halogen passivation would be unphysically overcrowsws.
The bonds face to the same missing carbon positions.
So these surfaces have to be
– either reconstructued and hydrogen passivated (slight periodic shifts)
– or passivated with (size matching) chalcogens (O,S,Se,(Te),((Po))) placed exactly at the place where the next layer of atoms would be.
The latter makes for parallel bridges averting snapback instabilities in the direction parallel to these bridges.
Atom size size, stability (chemical environment and mutual contact), toxicity and abundance considerations
all similar to the preceding section.
Pairings: (C,O),(Si,S),(Ge,Se),((Sn,Te))
Tellurium is extremely rare and quite metallic.
Polonium is even highly radioactive with a half life of 2898 years.
That beside diverging in chemical behavior from its group members above.
Cubic (110) faces
(wiki-TODO: Add section)
– These can be halogen passivated (particularly hydrogen passivated) without extreme hydrogen overcrowding.
… there is still some crowding that may cause some minute train.
– These are a bit smoother than halogen passivated cubic(111) faces.
– These can be passivated using pnictogens replacing the singly bond halogens by lone pairs
… averting snapback instabilities and possibly enabling repulsive Keesome forces from the strong dipole moment.
Surface passivation contact forces (exotic vdW forces)
Side-notes on exotic parts of vdW forcesforces:
To get repulsive Keesome forces from surface dipoles parts need to be guided so
they can't escape the undesired oppositions by rotating out of the way.
To get stronger attractive force than "normal" vdW forces
(just the virual dipole to virtual dipole london dispersion forces of e.g. two hydrogen passivated diamonds facing each other)
one can pair a nitrogen passivated surface with a hydrogen passivated surface.
I.e. dipol to induced dipole forces aka Debye forces.
These may be weak (TODO: Investigate the strencgt of such interactuons.)
Even stronger (attraqctive dipole to dipole keesome forces)
would require one surface with an opposite dipole moment.
But positive polarity i.e. boron (or even aluminum) passivation
is more questionable to achieve than the negative polarity.
Subsurface boron in slilicon is known and nonmechanosynthetically accessible.
A full boron layer on the top layer will remain to be seen if it can be mechanosynthesized.
Noter that a direct nitrogen to boron contact would make for extremely strong
datively bonding connection that is no longer a mobile interface.
like bulk sp3 boron nitrides being in the ballpark of bonding strenght of diamond.
Passivation of silicon containing gemstone like compounds
In the case of pure silicon the bigger size of silicon atoms already makes for more pronounced surface corrugations in case of a hydrogen passivation.
This makes designs for superlubricating surface interaction harder.
Increased corrugation already starts with moissanite (SiC) where only every second atom is a silicon.
A simple hydrogen passivation is less stable than in the case of carbon.
For silicon OH groups seem to be still quite stable.
As one can see from the highly stable Si-O bonds in polymer chains of current day available silicones.
The perhaps more stable hydroxy passivations are probably pretty bad for sliding interfaces because:
- Hydroxy passivations (·OH) are big singly bonded protrusions prone to snapback.
- With hydroxy passivations (·OH) being angled there is a unconstrained degree of freedom.
This may serve as an additional pathway for energy dissipation.
Surface corrugation really becomes terrible when going to sparsely filled oxide minerals like quartz (SiO2).
These have much bigger voids inside than more compact gemstone like compounds.
Plus they especially like hydroxy passivations.
Options for these more sparse materials:
- use them just for structural purposes
- give them some sort of much smoother surface cover like graphene maybe. Question is: How to "tack" it on neatly and tightly?
- look for similar less sparse materials stishovite instead of quartz (both polymorphs of Silicon dioxide SiO2
Passivations for transition metal monoxides
Maybe these could be covered with graphene?
See: Sandwich compound
Passivation by "graphene sheet lining"
It may be possible to passivate some base materials by tacking on graphene sheets onto the surface.
The bonds found in sandwich compound may be usable here.
Especially for materials that are otherwise hard to passivate this may be a possible option.
Graphene sheet lining may be an option for bigger sized gears
where teet are no longer single atoms but teeth instead already approximate evolvent or cycloid profiles.
Concerns:
- Can the tack-on density by high enough such that between the tack-ons there is not too low of a stiffness?
- Will a too dense tack-on pattern distort the graphenes electronic structure so much that it will become too reactive or even fully unstable?
- How well does the graphene conform to the underlying material?
- How much curvature is ok before localized kinks or too much change in electronic structure?
- How well can the graphene smooth out steps below in the underlying material?
- ... and so on and so forth ...
Intentionally incompatible material spacer shims
Two parts of a material that would like to fuse on contact
(like say e.g. rock salt structure TiC, TiN, TiO, or MgO)
could still by brought in sliding contact by putting in-between
a non-reacting "insulating" layer of hydrogen passivated diamond
or graphene or other suitable material.
To avoid having an positionally undefined (floating; out of machine phase) part
one can add some interdigitating from closure one one side.
Graphene would need some haptic bond pinning via metals on 5 rings of 5-7 dislocations.
Stone–Wales defect and
various variants of one or more combined azulene like patterns dislocations in graphene.
Related
- Chemical stability
- Macroscale surface passivation – Passivation layer mineral
- Passivation (disambiguation)
- Passivation bending issue – crystolecule deformations (strains) induced by undesired stresses caused by passivations
- Passivation layer mineral – This is about macroscale passivation. At the nanoscale passivation layer minerals still need passivation to not fuse together.
- Seamless covalent welding